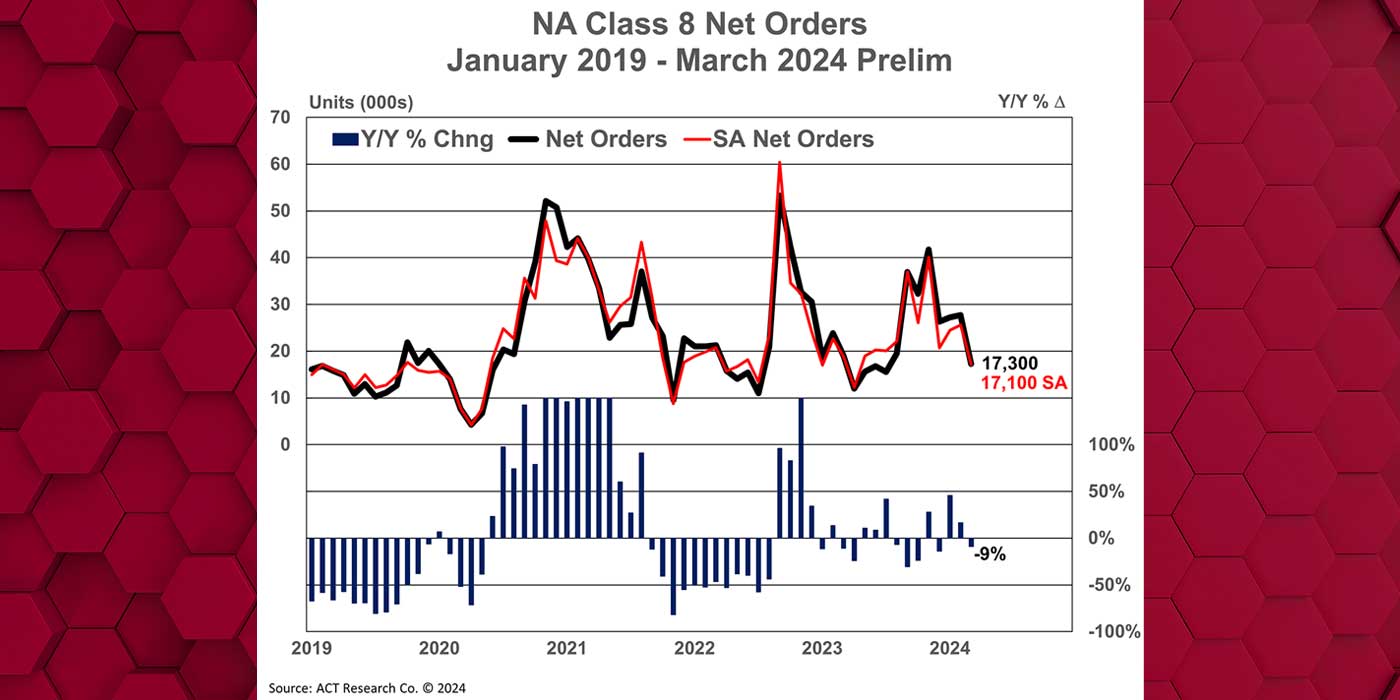
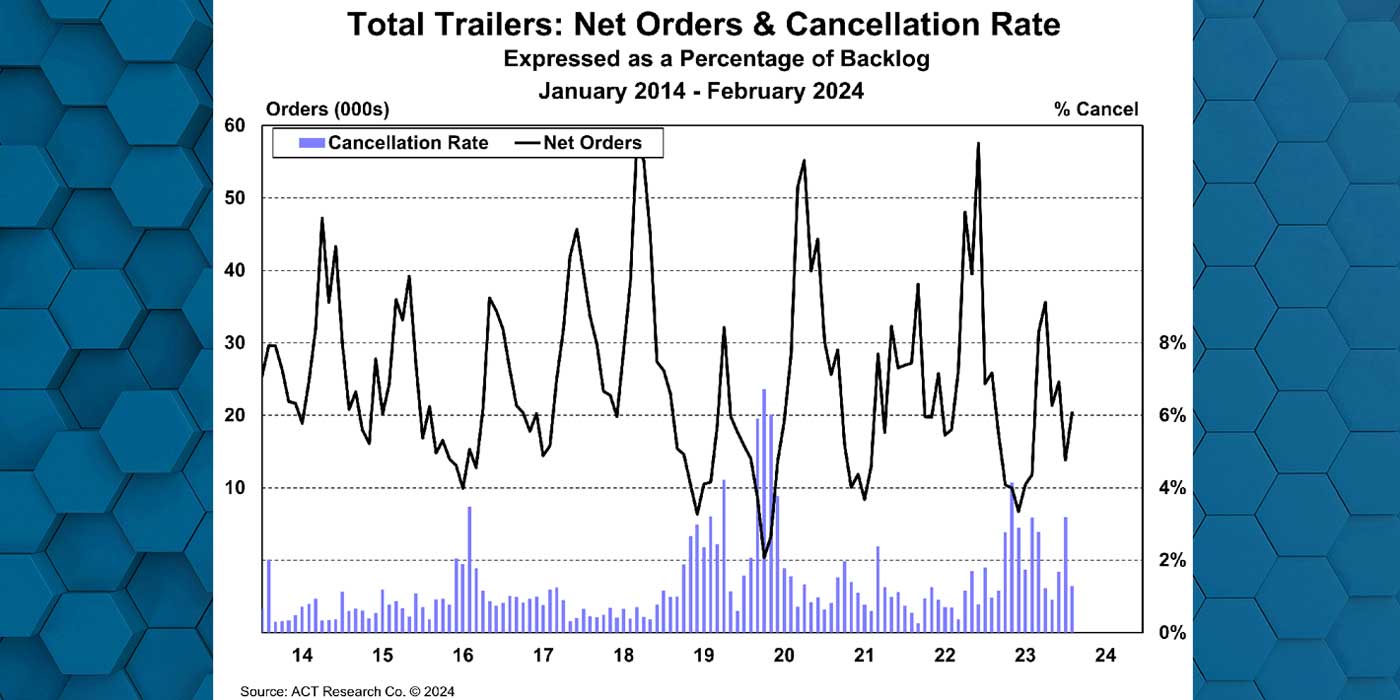
Thermo King has expanded its portfolio of temporary storage solutions that can meet the unique requirements of global pharmaceutical companies developing COVID-19 vaccines.
The company says pharmaceutical companies in final-stage clinical trials anticipate they will require strict temperature controls to safeguard their products—down to temperatures as low as -70 degrees Celsius. According to the World Health Organization, nearly 20% of temperature-sensitive health care products are damaged during transport, and 25% of vaccines reach their destination in a degraded state due to breaks in the cold chain.
Thermo King says it and its worldwide partners can offer temporary storage solutions that maintain a set point down to -70 degrees Celsius, and can ensure end-to-end temperature control, security and traceability through telematics. Additional storage solutions include refrigerated trailers, containers and portable cubes that can easily be scaled and repositioned to other locations as demand changes.
In addition to launching cold storage solutions, Thermo King says it has helped customers identify ways to maximize the range of dry ice, which is often used in vaccine transport and storage but has certain limitations. A container using dry ice to keep a product frozen may require re-icing if
it sits for an extended length of time or is exposed to extreme ambient weather. Thermo King offers storage solutions that it says can substantially extend the life of dry ice, or eliminate the need altogether.